Current & Emerging Treatments
Chronic graft-versus-host disease (cGVHD) is a long-term immune condition that can affect many parts of the body. Treatment usually occurs over a long period, with your care plan adapted as your condition changes. The main goals of treatment are to reduce symptoms and prevent permanent organ damage, preserve daily function and quality of life, limit side effects from medicines, and eventually allow the immune system to “calm down” so that immunosuppressive drugs can be safely reduced or stopped.1
Because cGVHD can involve the skin, eyes, mouth, lungs, joints, gut, and other organs, care is best provided by a multidisciplinary team (transplant specialists, organ specialists, nurses, rehabilitation therapists, pharmacists, social workers, and others).1
Nondrug and supportive care
Supportive care is a core part of cGVHD treatment and may help control symptoms, protect organs, and reduce the need for high-dose steroids.1
Examples include:
Eyes (ocular cGVHD)
Artificial tears, lubricating gels or ointments, punctal plugs to keep tears on the eye longer, moisture-chamber glasses, and special scleral lenses (such as PROSE lenses); early and regular visits with an eye specialist are important

Mouth and genital areas
Steroid mouth rinses or ointments, saliva-stimulating medicines (such as pilocarpine or cevimeline), and dilator therapy for genital scarring or tightness
Skin, fascia, and joints
Daily stretching, physical and occupational therapy, and deep-tissue massage for tight or hardened skin can maintain movement and prevent disability
Bone and metabolic health
Weight-bearing exercise (about 30 minutes at least 5 days per week), adequate calcium (~1500 mg/day) and vitamin D (~1000 IU/day), and sometimes medications such as bisphosphonates to prevent or treat bone loss
Infection prevention
Long-term antibiotics to prevent Pneumocystis pneumonia, medicines to prevent shingles (acyclovir or valacyclovir), blood tests to monitor for cytomegalovirus (CMV) and treat it early, and intravenous immune globulin (IVIG) for people with very low antibody levels

Adherence and emotional support
Clear medication education, help from pharmacists with complex schedules and drug interactions, cost and insurance navigation, counseling, and social work support
These approaches usually continue even when systemic (whole-body) medicines are started.
First-line systemic therapy: Corticosteroids
When cGVHD affects several organs or is moderate to severe, doctors usually start systemic corticosteroids (most often prednisone or methylprednisolone). These medicines broadly calm the immune system. Treatment often begins around 0.5 to 1 mg per kilogram of body weight per day. If the disease comes under control, the dose is slowly lowered over weeks to months. Many patients are tapered to about 0.2 to 0.25 mg/kg/day by 3 months if things are going well. Tapering is usually gradual, and often about a 20% to 30% dose reduction every 2 weeks, sometimes switching to every-other-day dosing. At each step, the team checks for early signs that cGVHD is flaring again.1,2
Because long-term steroids can cause serious side effects—such as bone loss, muscle weakness, high blood sugar or diabetes, high blood pressure, mood and sleep changes, cataracts, and higher infection risk—doctors try not to keep patients on high doses longer than necessary.1,2
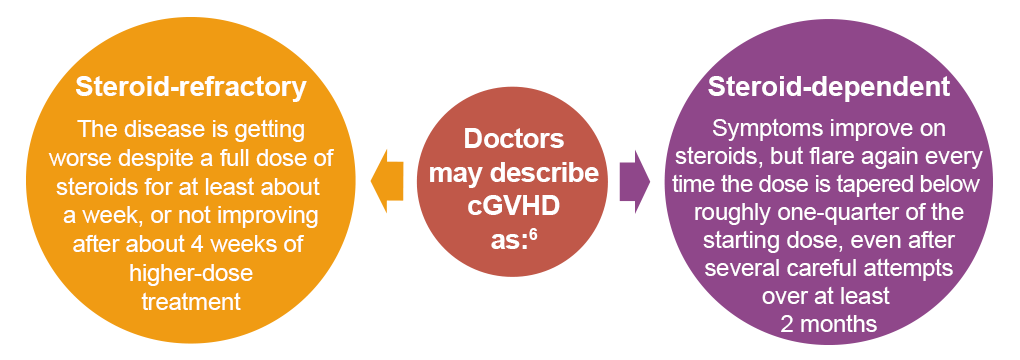
When steroids are no longer enough
Not everyone responds well to steroids. In fact, only about half of patients have long-lasting control with steroids alone, and about 50% to 60% need another systemic therapy within 2 years.1,2
These time points help the care team decide when it is safer and smarter to add or switch to another therapy instead of repeatedly increasing steroid doses.1,2
Targeted therapies for steroid-refractory or steroid-dependent cGVHD
For people whose cGVHD is steroid-refractory or steroid-dependent, several targeted therapies are now available. These medicines act on specific parts of the immune system or fibrosis (scarring) pathways. They are usually taken by mouth and often allow steroid doses to be reduced.2
The US Food and Drug Administration (FDA) has currently approved 4 oral medicines in the United States for steroid-refractory cGVHD2:
Ibrutinib (BTK/ITK inhibitor)
Ibrutinib blocks proteins (Bruton tyrosine kinase [BTK] and interleukin-2 inducible T-cell kinase [ITK]) that help B and T cells stay activated.2 Clinical trials showed an overall response rate (ORR) around 67%, with improvement in skin, mouth, and digestive tract symptoms.3 Real-world studies show responses in about 45% of patients at 6 months, with especially good responses in some patients with liver involvement.4
Possible side effects
Fatigue, diarrhea, easy bruising or bleeding, low blood counts, and infections; because it is broken down by the liver enzyme CYP3A4, many other medicines (such as certain antifungals or antibiotics) can interact with it so your team may adjust doses or choose alternatives to avoid dangerous interactions2
Ruxolitinib (JAK 1/2 inhibitor)
Ruxolitinib blocks Janus kinase–signal transducer and activator of transcription (JAK-STAT) signaling, a pathway that carries many inflammatory signals.2 In the large REACH3 trial, ruxolitinib achieved an ORR of about 50% at 24 weeks, nearly doubling the responses seen with the “best available” standard therapies. Responses occurred across skin, mouth, gut, and lung involvement, and many patients were able to lower their steroid dose.5 Longer-term follow-up shows longer failure-free survival and more durable responses than standard treatments.6
Possible side effects
Low red blood cells, white cells, or platelets (cytopenias); infections including viral reactivation; and increases in cholesterol or fats in the blood so regular blood tests and infection monitoring are essential2
Belumosudil (ROCK2 inhibitor)
Belumosudil blocks Rho-associated coiled-coil containing protein kinase 2 (ROCK2), affecting both immune balance and fibrosis.2 In the ROCKstar trial, heavily pretreated patients had an ORR around 74%, with many responding within about 5 weeks and a median steroid dose reduction of ~45%.7 Longer-term follow-up (about 2.5 years) showed responses remaining durable (ORR ~72%) with no new safety concerns.8 Belumosudil is particularly attractive for fibrotic skin, fascia, or joint disease, in which tightness and limited movement are major issues.7
Axatilimab (CSF-1R inhibitor)
Possible side effects
Mostly gastrointestinal (nausea, diarrhea) and constitutional (fatigue, cough); overall, it is generally well-tolerated, but you will still need regular monitoring2
Axatilimab targets colony-stimulating factor (CSF-1) receptors on monocytes and macrophages, immune cells that can drive inflammation and scarring.2 In the AGAVE-201 trial, which enrolled heavily pretreated patients, the ORR was about 74%.
Possible side effects
Fatigue and elevations in certain blood tests—liver enzymes (aspartate transaminase [AST], alanine transaminase [ALT]), muscle enzyme (creatine phosphokinase [CPK}), lactate dehydrogenase (LDH), and lipase so regular lab monitoring is required, and doses may be adjusted if levels rise2
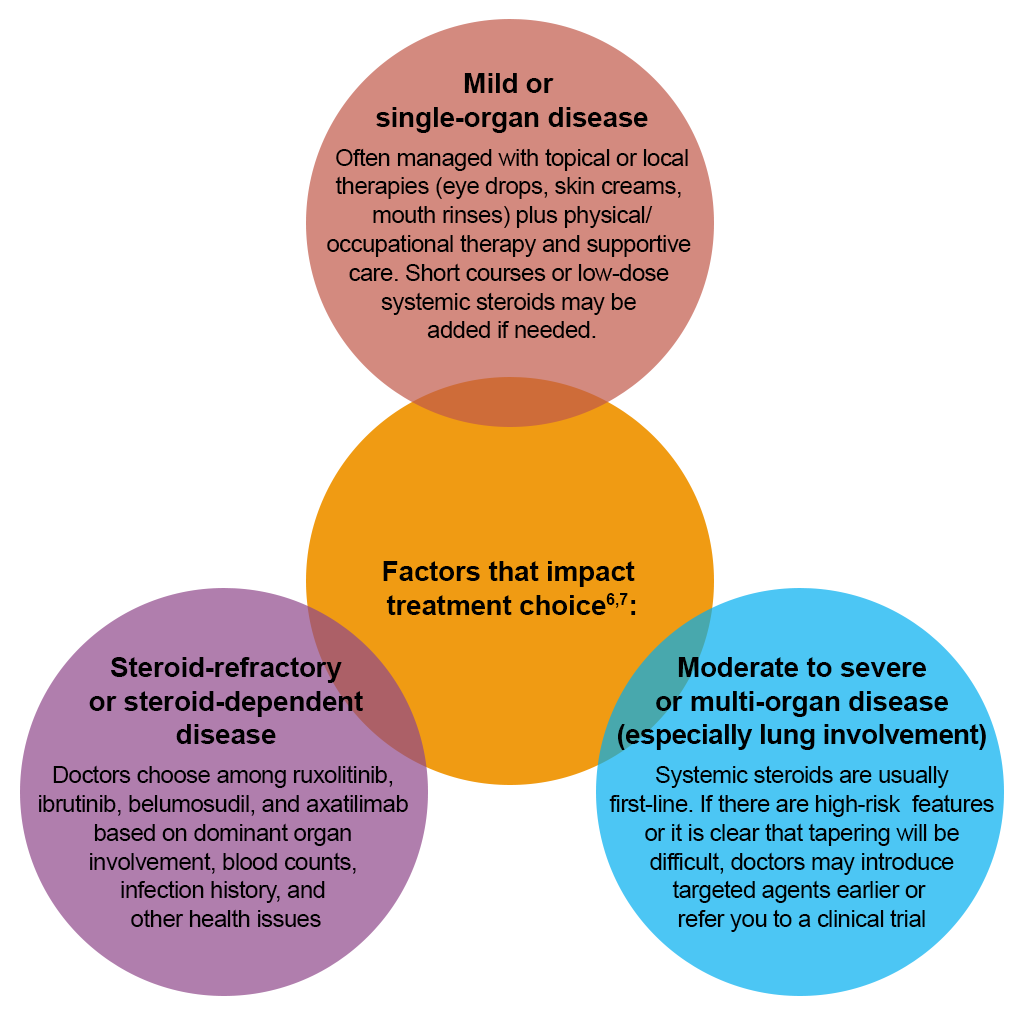
How doctors choose among treatments
No single treatment is right for everyone. Your team considers many factors when designing or adjusting your plan, including which organs are affected and how severe they are, past treatments and responses, other health conditions, and your goals and preferences.1,2
Examples of organ-directed choices
The following general patterns can help explain why your team might recommend 1 medicine over another2:
- Inflammatory skin, mouth, or gut disease
Ruxolitinib or ibrutinib are often considered early second-line options; belumosudil can be added later, especially if there is a mix of inflammation and fibrosis - Fibrotic skin, fascia, or joint involvement
Belumosudil is commonly favored because of its antifibrotic effects; extracorporeal photopheresis (ECP)—a procedure where blood cells are treated with light-activated medicine and returned to the body—may also help, and physical and occupational therapy remain critical - Lung involvement
Ruxolitinib has shown meaningful lung responses in clinical trials and real-world studies; it may be combined with inhaled medicines, certain antibiotics (such as macrolides), and pulmonary rehabilitation - Heavily pretreated, multiorgan disease
Belumosudil, axatilimab, or combinations may be used, and clinical trial enrollment is encouraged when available; other options in selected cases include ECP, rituximab, mammalian target of rapamycin (mTOR) inhibitors, or mesenchymal stromal cell therapy
Emerging and investigational treatments
Research in cGVHD is moving quickly. Clinical trials are exploring whether these newer medicines should be used earlier in treatment, instead of waiting until steroids no longer work. Several clinical trials are testing combinations of steroids and targeted drugs—or even steroid-free approaches from the start. These studies are designed to learn whether starting targeted therapy sooner can improve long-term outcomes and reduce the need for long-term steroid use. The results of these trials may change how cGVHD is treated in the future.9
Participating in a clinical trial may give access to promising therapies and helps improve care for future patients. Your transplant team can tell you which trials might be a good fit, based on your disease features and treatment history.10
References
- Flowers MED, Martin PJ. How we treat chronic graft-versus-host disease. Blood. 2015;125(4):606-615. https://doi.org/10.1182/blood-2014-08-551994
- Vadakkel G, Eng S, Proli A, Ponce DM. Updates in chronic graft-versus-host disease: novel treatments and best practices in the current era. Bone Marrow Transplant. 2024;59(10): 1360-1368. https://doi.org/10.1038/s41409-024-02370-8
- Ibrutinib (Imbruvica®) Prescribing information (PI) 2025. https://www.imbruvicahcp.com/cgvhd/efficacy/overall-response-rate Accessed 12/14/2025.
- Pidala J, Kim J, Kalos D, et al. Ibrutinib for therapy of steroid-refractory chronic graft-versus-host disease: a multicenter real-world analysis. Blood Adv. 2025;9(5):1040-1048. https://doi.org/10.1182/bloodadvances.2024014374
- Zeiser R, Polverelli N, Ram R, et al. Ruxolitinib for glucocorticoid-refractory chronic graft-versus-host disease. N Engl J Med. 2021;385(3):228-238. https://doi.org/10.1056/NEJMoa2033122
- Mohty M, Socié G, Szer J, et al. Ruxolitinib versus best available therapy in patients with steroid-refractory acute graft-versus-host disease: final analysis from the randomized phase III REACH2 trial. J Clin Oncol. 2025;43(34):3639-3645. https://doi.org/10.1200/JCO-25-00809
- Cutler C, Lee SJ, Arai S, et al. Belumosudil for chronic graft-versus-host disease after 2 or more prior lines of therapy: the ROCKstar Study. Blood. 2021;138(22):2278-2289. https://doi.org/10.1182/blood.2021012021
- Lee SJ, Pavletic S, Blazar BR, et al. Belumosudil for chronic graft-versus-host disease: analysis of long-term results from the KD025-208 and ROCKstar studies. Transplant Cell Ther. 2025;31(7):434.e1-434.e10. https://doi.org/10.1016/j.jtct.2025.04.020
- Salhotra A. Emerging treatment practices in cGvHD care. 12/10/2025. https://www.targetedonc.com/view/emerging-treatment-practices-in-cgvhd-care Accessed 12/14/2025.
- National Comprehensive Cancer Network (NCCN). Graft-versus-host disease. 2025. https://www.nccn.org/guidelines/guidelines-detail?category=patients&id=63. Accessed 12/14/2025.