cGVHD Pathophysiology
What is chronic graft-versus-host disease?
Chronic graft‑versus‑host disease (cGVHD) is a late complication of allogeneic hematopoietic cell transplantation in which donor immune cells mount a persistent, dysregulated response against host tissues, resulting in chronic inflammation, tissue damage, and progressive fibrosis across multiple organs. It is now understood as an immune‑mediated, autoimmune‑like disorder rather than simply “late acute” GVHD, with complex interactions among T cells, B cells, and innate immune cells driving heterogeneous clinical phenotypes.1-3
Biological phases of cGVHD
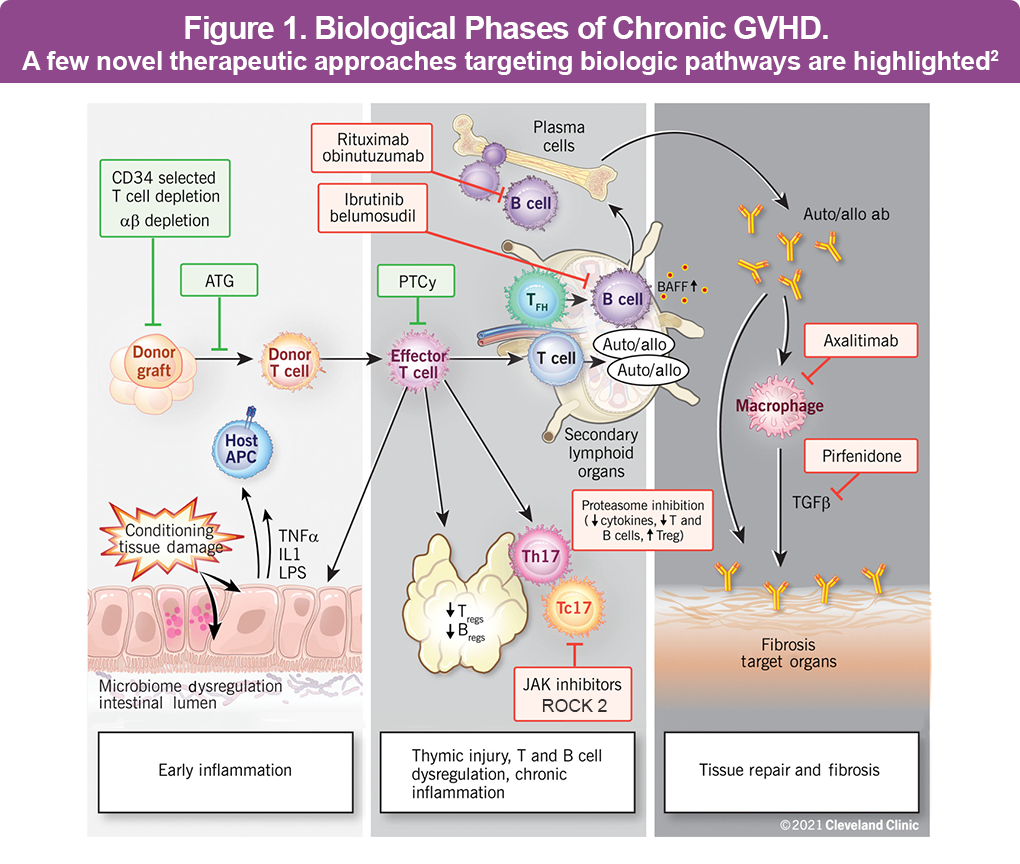
Chronic GVHD develops through overlapping biological phases that begin early after transplant, often before clinical signs are apparent. Although the underlying pathogenesis of cGVHD is not fully understood, a widely used model describes 3 major phases: early inflammation and tissue injury, immune dysregulation, and aberrant tissue repair with fibrosis (Figure 1).2,3
- Phase 1 – Early inflammation and tissue injury: Conditioning and prior acute GVHD damage epithelial barriers and parenchymal tissues, releasing danger signals and upregulating antigen‑presenting cell (APC) function, with increased inflammatory cytokines such as interleukin (IL)‑1, tumor necrosis factor (TNF), and IL‑6.1
- Phase 2 – Immune dysregulation: Thymic injury impairs central tolerance, regulatory T cells are reduced or dysfunctional, and T‑follicular helper and B‑cell germinal center responses become exaggerated, promoting alloantibody and autoantibody production.1
- Phase 3 – Aberrant tissue repair and fibrosis: Persistent activation of macrophages and fibroblasts—driven by mediators including transforming growth factor-beta (TGF‑β) and platelet-derived growth factor (PDGF)—leads to excessive extracellular matrix deposition and scarring, manifesting as sclerotic skin changes, joint/fascia involvement, and bronchiolitis obliterans, among others.1
These phases may occur sequentially or overlap within a given organ, and not all patients pass distinctly through each phase, which contributes to the clinical heterogeneity of cGVHD.1
Key immune and stromal players
Multiple immune and stromal cell populations participate in cGVHD pathobiology. Donor CD4+ and CD8+ T cells recognize host alloantigens presented by APCs and differentiate into a spectrum of effector phenotypes, including Th1, Th2, and particularly Th17/Tc17 subsets that secrete IL‑17, IL‑21, interferon gamma (IFN‑γ), and other cytokines sustaining chronic inflammation. B‑cell abnormalities include loss of naïve and regulatory B cells, elevated B-cell activating factor (BAFF) levels, and production of alloantibodies and autoantibodies, which can activate endothelial and mesenchymal cells and correlate with sclerotic disease (Figure 1).2,3
Innate immune cells, especially colony-stimulating factor 1 (CSF‑1)–dependent macrophages, serve as critical effectors in later fibrotic stages by producing TGF‑β and PDGF that drive fibroblast activation, collagen deposition, and tissue stiffening. Fibroblasts and myofibroblasts themselves become persistently activated, generating and cross‑linking extracellular matrix proteins that convert reversible inflammatory lesions into fixed organ damage and functional impairment (Figure 1).2,3
The central role of chronic inflammation
Chronic inflammation is both a driver and a consequence of ongoing immune dysregulation in cGVHD. Persistent low-grade inflammatory signals are maintained by microbial products, tissue damage, dysregulated cytokine networks, and autoantibodies, which promote continuous recruitment and activation of effector T cells, B cells, and macrophages in target organs. Over time, this smoldering inflammation transitions from predominantly cellular infiltrates and epithelial damage to dense fibrosis, atrophy, and organ dysfunction, explaining the evolution from inflammatory to sclerotic clinical phenotypes.1-3
Steroid-responsive cGVHD
Systemic corticosteroids—typically prednisone with or without a calcineurin inhibitor—are the recommended first‑line therapy for moderate to severe cGVHD. Steroid-responsive cGVHD is characterized by clear clinical improvement or resolution of signs and symptoms on standard-dose prednisone and the ability to taper steroids, often below approximately 0.25 mg/kg/day, without disease flare. Approximately 50% of patients respond to initial therapy, with the remaining developing either steroid-refractory or steroid-dependent disease.4
Steroid-refractory and steroid-dependent cGVHD
Steroid‑refractory (SR) cGVHD is defined as 1 or more of the following: disease progression while receiving prednisone ≥1 mg/kg/day for 1 to 2 weeks; stable disease without improvement despite prednisone ≥0.5 mg/kg/day for 1 to 2 months; or steroid‑dependent disease with recurrent flares when prednisone is tapered below about 0.25 mg/kg/day (Table 1). Up to half or more of patients with moderate to severe cGVHD ultimately become steroid‑refractory or steroid‑dependent and require second‑line systemic therapies.4 Prolonged corticosteroid therapy is associated with a range of adverse effects, including osteoporosis, myopathies, mood disturbances, weight gain, hypertension, and diabetes. These challenges underscore the necessity for more effective, safer, and targeted treatments (such as Janus kinase [JAK], Bruton tyrosine kinase [BTK], and Rho-associated kinase 2 [ROCK2] inhibitors) that can selectively modulate cGVHD pathways with reduced toxicity.4
References
Flavin B. Chronic graft-vs-host disease: current understanding of disease and treatment landscape. J Manag Care Spec Pharm. 2022;28(suppl 12-bl):S2-S12. https://doi.org/10.18553/jmcp.2022.28.12-b.s1
Hamilton BK. Updates in chronic graft-versus-host disease. Hematology. 2021;2021(1):648-654. https://doi.org/10.1182/hematology.2021000301
Cooke KR, Luznik L, Sarantopoulos S, et al. The biology of chronic graft-versus-host disease: a task force report from the National Institutes of Health Consensus Development Project on criteria for clinical trials in chronic graft-versus-host disease. Biol Blood Marrow Transplant. 2017;23(2):211-234. https://doi.org/10.1016/j.bbmt.2016.09.023
Vadakkel G, Eng S, Proli A, Ponce DM. Updates in chronic graft-versus-host disease: novel treatments and best practices in the current era. Bone Marrow Transplant. 2024;59(10): 1360-1368. https://doi.org/10.1038/s41409-024-02370-8
- National Comprehensive Cancer Network (NCCN). Hematopoietic cell transplantation. Version 2.2025. https://www.nccn.org/guidelines/guidelines-detail?category=3&id=1501 Accessed 12/13/2025.