Diagnosing cGVHD
The diagnosis of cGVHD is based on specific clinical manifestations rather than the time since transplant. Previously, GVHD present after 100 days posttransplant was classified as chronic, but advances in transplant practices have shown that acute and chronic GVHD can overlap in timing and presentation. As a result, current guidelines rely on clinical features, rather than onset timing, to distinguish chronic from acute GVHD.1
How cGVHD is diagnosed
Chronic GVHD is diagnosed when a patient has1:
- At least 1 diagnostic manifestation, which alone is sufficient to establish the diagnosis, or
- At least 1 distinctive manifestation, confirmed by biopsy, laboratory testing, or imaging in the same or another organ.
Importantly, other causes such as infection, medication toxicity, or malignancy must always be excluded before confirming the diagnosis.1
Key organ systems involved
Chronic GVHD can affect multiple organs, most commonly:
Skin and appendages
(eg, sclerotic changes, lichen-like features, poikiloderma)
Mouth
(eg, lichen-type changes, mucosal atrophy, ulcers)
Female genital tract
(eg, scarring, stenosis, erosions
Gastrointestinal tract and esophagus
(eg, strictures, pancreatic insufficiency)
Liver
(elevated bilirubin or liver enzymes)
Lungs
(bronchiolitis obliterans confirmed by biopsy, imaging, or pulmonary function tests)
Joints, fascia, and muscles
(fasciitis, contractures, myositis)
Some manifestations are diagnostic, some are distinctive but require confirmation, while others are common to both acute and chronic GVHD and cannot be used alone to establish the diagnosis.1
Role of biopsy and testing
Biopsy, imaging, and specialized tests (such as pulmonary function tests or Schirmer testing for the eyes) are strongly encouraged to confirm the diagnosis, especially when only distinctive features are present. However, biopsy is not mandatory if a clear diagnostic clinical feature is identified.1
Diagnosis vs severity assessment
Features used to establish the diagnosis of cGVHD are not always suitable for assessing disease severity or treatment response. Diagnostic criteria prioritize accuracy, while response measures prioritize sensitivity to change—meaning separate tools are required for diagnosis and for monitoring treatment effectiveness.1
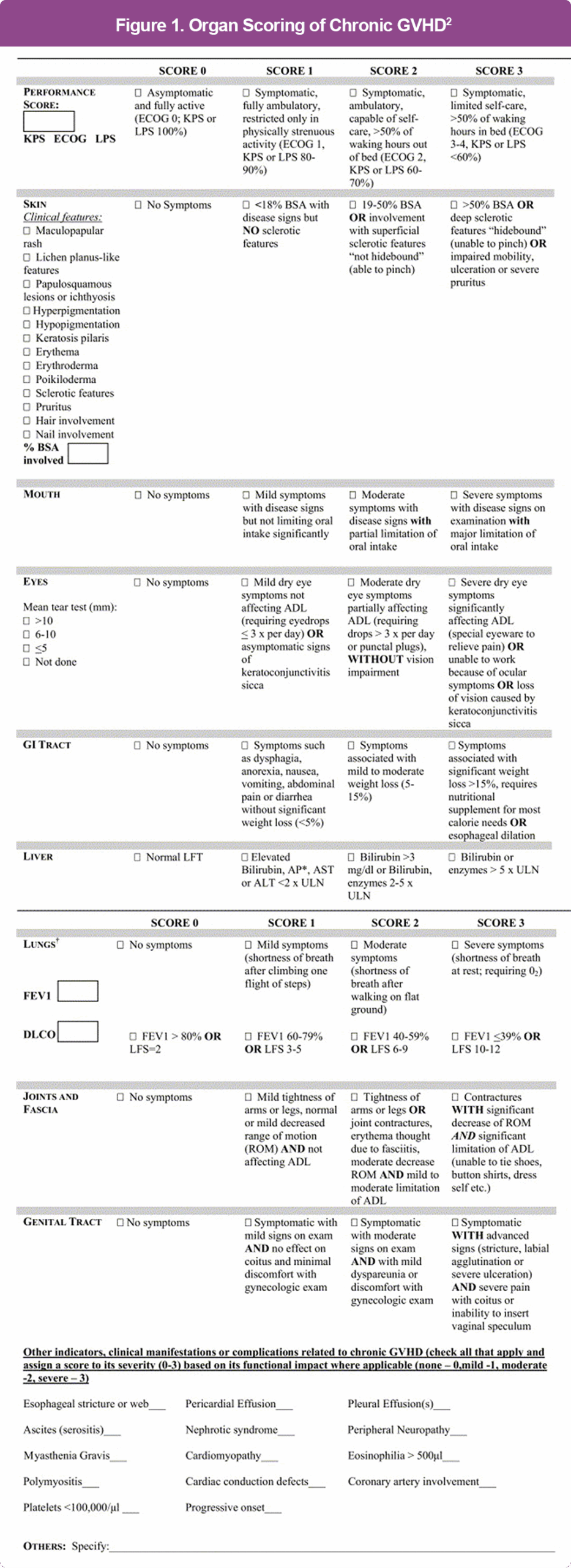
Grading cGVHD
Grading of cGVHD follows the National Institutes of Health (NIH) Consensus Development Project criteria using an organ-based scoring system that evaluates the skin, mouth, eyes, gastrointestinal tract, liver, lungs, joints and fascia, and female genital tract. Each site is rated on a 0 to 3 scale, where 0 indicates no involvement and 3 indicates severe impairment. Performance status is also scored from 0 to 3, with additional checkboxes used to document other specific manifestations. Importantly, the assessment (Figure 1) is based solely on the patient’s current clinical status, without regard to prior findings or the need to distinguish cGVHD-related abnormalities from preexisting conditions.1
Early recognition of cGVHD
Early recognition of cGVHD remains a critical unmet need, as many patients do not meet NIH diagnostic criteria until organ damage is already underway. Despite advances in standardizing definitions and improving trial design, the real-world implementation of diagnostic guidelines is inconsistent. A future-focused approach will require coordinated efforts among transplant specialists, community clinicians, patients, and caregivers to detect subtle, prediagnostic signs before irreversible manifestations develop. Longitudinal studies beginning at or before transplant—incorporating serial clinical evaluations, patient-reported outcomes, biomarkers, and emerging technologies such as handheld spirometry and advanced imaging— are essential to establish reliable early indicators of disease. Ultimately, validating these early features will pave the way for preemptive intervention strategies that can reduce morbidity, preserve organ function, and improve long-term outcomes for hematopoietic cell transplant survivors.3
References
Filipovich AH, Weisdorf D, Pavletic S, et al. National Institutes of Health Consensus Development Project on criteria for clinical trials in chronic graft-versus-host disease: I. Diagnosis and Staging Working Group Report. Biol Blood Marrow Transplant. 2005;11(12):945-956. https://doi.org/10.1016/j.bbmt.2005.09.004
Jagasia MH, Greinix HT, Arora M, et al. National Institutes of Health Consensus Development Project on criteria for clinical trials in chronic graft-versus-host disease: I. The 2014 Diagnosis and Staging Working Group Report. Biol Blood Marrow Transplant. 2015;21(3):389-401.e1. https://doi.org/10.1016/j.bbmt.2014.12.001
Kitko CL, Pidala J, Schoemans HM, et al. National Institutes of Health Consensus Development Project on criteria for clinical trials in chronic graft-versus-host disease: IIa. The 2020 Clinical Implementation and Early Diagnosis Working Group Report. Transplant Cell Ther. 2021;27(7): 545-557. https://doi.org/10.1016/j.jtct.2021.03.033