Treating cGVHD
Chronic graft-versus-host disease (cGVHD) is a chronic, multisystem immune-mediated disorder that requires long-term, individualized management. The goals of treatment are to:
Reduce symptom burden and prevent irreversible damage
Preserve function and quality of life
Minimize treatment-related toxicity
Ultimately achieve immune tolerance and safely withdraw immunosuppression
Because cGVHD affects many organs and dimensions of life, optimal care is multidisciplinary and integrates nonpharmacologic strategies, systemic therapies (steroids and targeted agents), and proactive adverse event management.1
Nonpharmacologic and supportive approaches
Supportive care is essential throughout the disease course and may reduce steroid exposure, improve function, and prevent disability1:
Ocular cGVHD
Artificial tears, punctal plugs or ligation, moisture-chamber eyewear, PROSE scleral lenses, and early ophthalmology comanagement
Oral/genital cGVHD
Topical steroid rinses/ointments, dilator therapy, and sialagogues (pilocarpine, cevimeline)
Skin, fascia, and joints
Daily stretching, physical and occupational therapy, deep-tissue massage for sclerotic disease
Bone and metabolic health
o Weight-bearing exercise ≥30 minutes, ≥5 days/week
o Calcium ~1500 mg/day, vitamin D ~1000 IU/day
o Consider bisphosphonates for osteopenia
Infection prevention
Long-term pneumocystis and antibacterial prophylaxis, varicella-zoster virus prophylaxis with acyclovir/valacyclovir, cytomegalovirus surveillance with preemptive therapy, intravenous immune globulin for hypogammaglobulinemia
Adherence and psychosocial support
Medication education, pharmacist-led counseling, cost navigation, and social work support
First-line systemic therapy: Corticosteroids
Corticosteroids remain the backbone of initial systemic treatment. Prednisone or methylprednisolone is typically initiated at 0.5 to 1 mg/kg/day, followed by gradual tapering once disease control is achieved. In prospective studies, prednisone is often reduced to ~0.2 to 0.25 mg/kg/day within 3 months.1,2
Tapering is highly individualized but generally follows these principles1,2:
- Progressive reduction of ~20% to 30% every 2 weeks
- Transition to alternate-day dosing as tolerated
- At each step, patients must be examined for early disease recurrence
- If GVHD flares, the steroid dose is escalated promptly and maintained for several weeks before another taper attempt
When does disease become steroid-refractory or steroid-dependent?
Despite careful tapering, only about 50% of patients achieve durable control with steroids alone, and 50% to 60% require second-line therapy within 2 years.1,2
Definitions: 2
- Steroid-refractory cGVHD
- Progression on prednisone 1 mg/kg/day for ≥7 days, or
- No improvement on ≥0.5 mg/kg/day for ≥4 weeks
- Steroid-dependent cGVHD
- Inability to taper below ~0.25 mg/kg/day after ≥2 failed taper attempts separated by ≥8 weeks
These definitions and time anchors (1 week, 4 weeks, 2 months of high doses, repeated failed taper attempts) guide when to transition to secondary systemic therapy rather than indefinitely escalating steroids.1,2
Long-term steroid exposure is associated with osteoporosis, myopathy, diabetes, hypertension, mood disturbance, cataracts, and infection. Thus, an earlier transition to targeted therapies when appropriate is ideal.2
Targeted therapies for steroid-refractory/steroid-dependent cGVHD
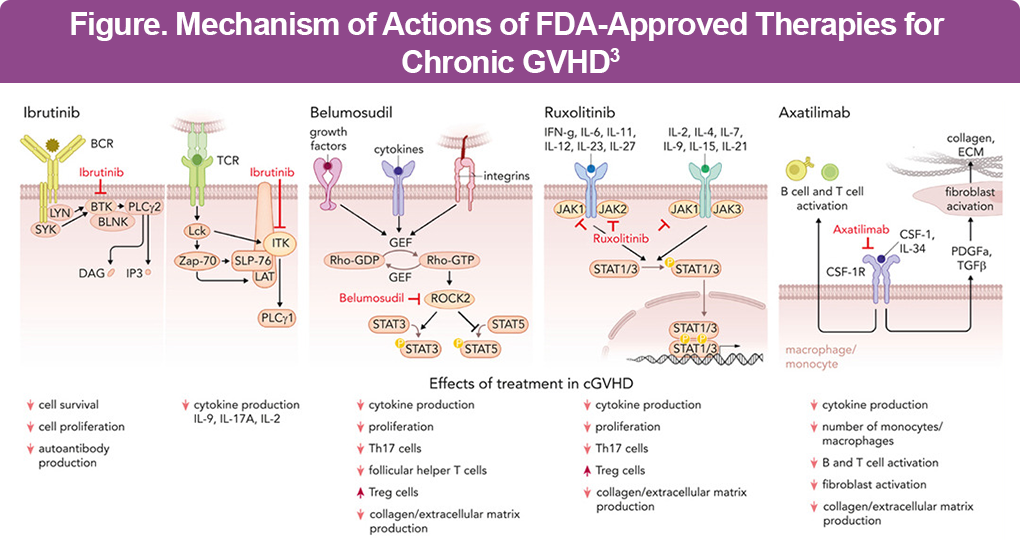
Once cGVHD is steroid-refractory or steroid-dependent, targeted agents play a key role. The US Food and Drug Administration (FDA) has approved 4 oral agents for steroid-refractory cGVHD, each targeting distinct immune and fibrotic pathways (Figure).2,4
BTK/ITK inhibition – ibrutinib
Ibrutinib irreversibly inhibits Bruton tyrosine kinase (BTK) in B cells and interleukin-2 inducible T-cell kinase (ITK) in T cells, suppressing antigen-driven activation, survival, and cytokine signaling.4 In clinical trials, the overall response rates (ORR) approached 67%, with notable activity in the skin, mouth, and gastrointestinal (GI) tract.4 A real-world study showed that ibrutinib achieved a 6-month ORR of 45%, while liver involvement predicted better response and older age, higher baseline prednisone, and lung involvement were associated with worse failure-free survival, with discontinuation driven mainly by progression or toxicity.5
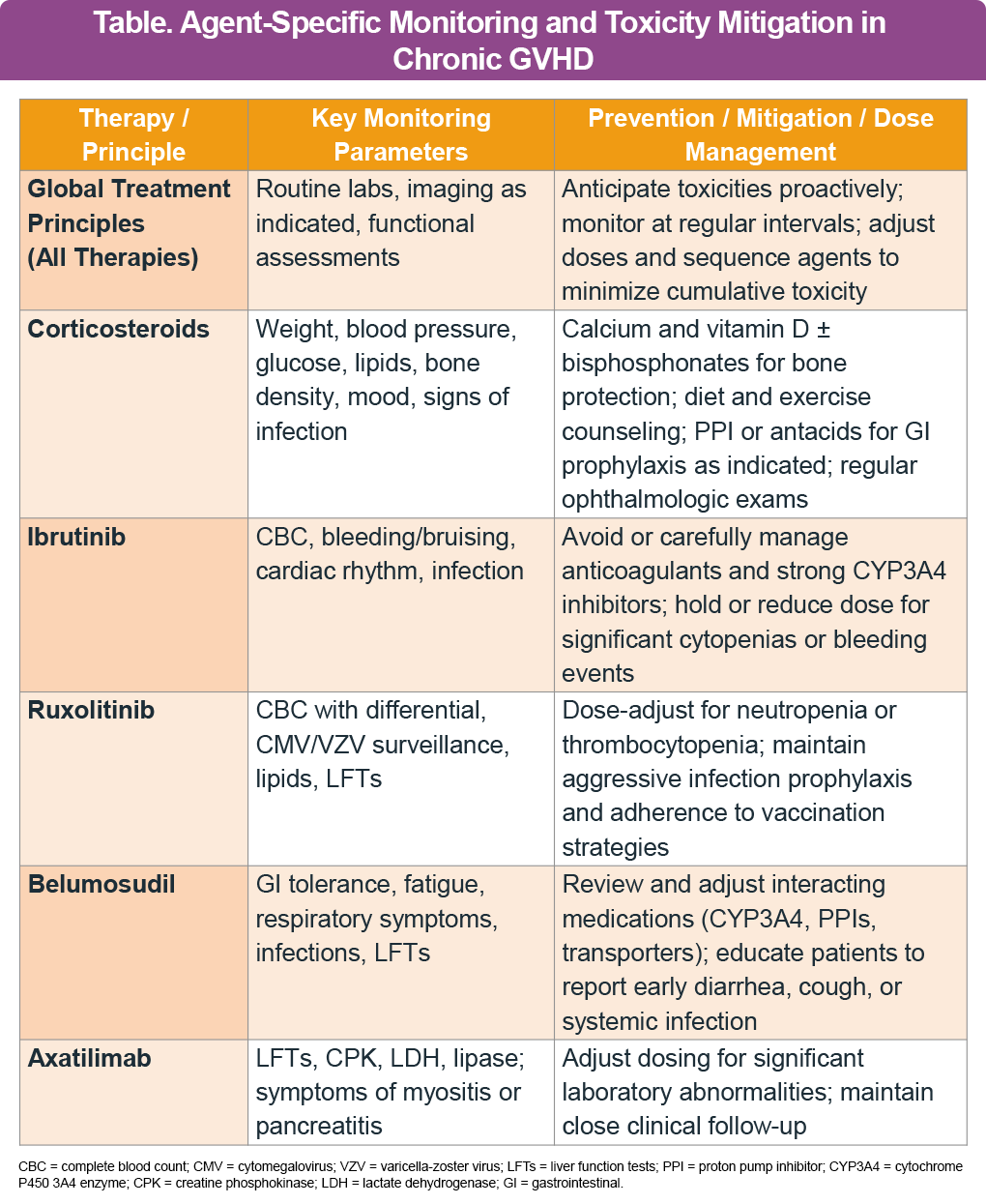
Common adverse events (AEs) include fatigue, diarrhea, bruising, cytopenias, and infection, with important drug–drug interactions via CYP3A4.4
ROCK2 inhibition – belumosudil
Belumosudil selectively inhibits Rho-associated kinase 2 (ROCK2), exerting both immunomodulatory and antifibrotic effects. It shifts the immune balance away from Th17-driven inflammation toward regulatory T-cell activity while also suppressing profibrotic gene expression and collagen deposition.2
In the ROCKstar trial of heavily pretreated patients, the ORR was ~74%, with a median time to response of ~5 weeks and substantial steroid-sparing (median dose reduction ~45%). Belumosudil has been noted to be particularly effective for patients with fibrotic skin, fascia, and joint disease.6 Long-term results (overall median follow-up duration of 31.4 months) showed that belumosudil was associated with durable responses (ORR of 72%) and remained well-tolerated with no new safety concerns.7
AEs are primarily GI and constitutional (fatigue, diarrhea, nausea, cough), and overall tolerability is favorable.4
JAK1/2 Inhibition – ruxolitinib
Ruxolitinib blocks Janus kinase-signal transducer and activator of transcription (JAK-STAT) signaling, thereby suppressing proinflammatory cytokine signaling and alloreactive T-cell trafficking into target organs.2 In the phase 3 REACH3 trial, ruxolitinib achieved an ORR of ~50% at 24 weeks, nearly doubling responses compared with best available therapy. Responses occur across skin, oral, GI, and lung involvement, and real-world data confirm meaningful steroid-sparing effects.8 In the 3-year final analysis, ruxolitinib was associated with longer failure-free survival and duration of response than best available therapy in patients with steroid-refractory or steroid-dependent cGVHD.9
Key AEs include cytopenias, infections (including viral reactivation), and hyperlipidemia, requiring close laboratory and infection surveillance.2
CSF-1R inhibition – axatilimab
Axatilimab targets colony-stimulating factor-1 receptor (CSF-1R) on pathogenic monocytes and macrophages, disrupting macrophage-driven inflammation and fibrosis. In the phase 2 AGAVE-201 trial, the ORR reached ~74% in heavily pretreated patients.10
Adverse events reflect macrophage and tissue enzyme effects and include fatigue and elevations of aspartate aminotransferase (AST), alanine aminotransferase (ALT), creatine phosphokinase (CPK), lactate dehydrogenase (LDH), and lipase, necessitating routine laboratory monitoring.
Aligning treatment selection with organ involvement and severity10
Treatment selection is not 1-size-fits-all. It should integrate NIH organ scoring (mild/moderate/severe), pattern of involvement (inflammatory vs fibrotic; skin vs lung vs gastrointestinal, etc), prior lines and responses, and comorbidities, cytopenias, infection history, and drug–drug interactions.10
General framework
Management of cGVHD is guided by disease severity, organ involvement, and treatment response. Patients with mild, limited, or single-organ involvement can often be managed with topical or local therapies, physical or occupational therapy, and supportive care, with short courses or low-dose systemic corticosteroids used selectively when symptoms are not adequately controlled.
In patients with moderate to severe multiorgan disease or lung involvement, systemic corticosteroids remain first-line therapy, with early consideration of clinical trial enrollment and/or targeted agents in those with high-risk features or disease that is difficult to taper.
For steroid-refractory or steroid-dependent cGVHD, selection among ruxolitinib, ibrutinib, belumosudil, or axatilimab should be individualized based on the dominant organ pattern (eg, fibrotic skin and joint disease vs inflammatory GI or pulmonary involvement), cytopenia profile (with preference for belumosudil or axatilimab in the setting of significant cytopenias and cautious use of ruxolitinib), infection history (particularly recurrent viral reactivation with JAK inhibition), and relevant comorbidities and drug–drug interactions, including atrial fibrillation, anticoagulation, azole antifungals, and hepatic dysfunction.10
Examples of organ-directed systemic choices (conceptual)
- Predominantly inflammatory skin/oral/GI disease
- Ruxolitinib or ibrutinib often considered early second line
- Belumosudil can be effective in more heavily pretreated patients or mixed inflammatory/fibrotic phenotypes
- Fibrotic skin, fascia, or joint involvement
- Belumosudil and extracorporeal photopheresis (ECP) commonly favored due to antifibrotic and Treg-promoting effects
- Physical therapy and deep tissue massage are essential adjuncts
- Lung involvement
- Ruxolitinib has meaningful lung responses in trials and real-world data
- Consider combinations with inhaled therapies, macrolides, and pulmonary rehabilitation
- Heavily pretreated, multiorgan, refractory disease
- Belumosudil, axatilimab, and/or enrollment in clinical trials
- Extracorporeal photophoresis, rituximab, mammalian target of rapamycin (mTOR) inhibitors, or mesenchymal stromal cells as situation-specific options
Adverse events and their management across therapies
Treatment of cGVHD requires proactive toxicity anticipation, regular multidisciplinary monitoring (including laboratory studies, imaging, and functional assessments), and thoughtful dose adjustment and sequencing to limit cumulative adverse effects.1,2
Overcoming barriers to effective cGVHD treatment
Effective treatment of cGVHD is frequently challenged by several practical barriers, including polypharmacy and high pill burden—contributing to nonadherence in patients with moderate to severe disease—along with the high costs of targeted agents, insurance authorization hurdles, limited access to cGVHD specialists and coordinated multidisciplinary care, and patient concerns regarding tolerability and potential adverse effects.
Strategies to overcome these barriers include simplifying regimens and consolidating dosing schedules whenever feasible, engaging clinical pharmacists to provide regular education and adherence support, leveraging social work and financial counseling services to mitigate cost and access issues, supplying patients with clear written treatment plans that outline action steps for disease flares and adverse events, and encouraging participation in clinical trials for eligible patients to expand access to emerging therapies.2
Adverse events and their management across therapies
The management of cGVHD requires an ongoing balance between effective disease control, toxicity minimization, and the long-term goal of achieving immune tolerance. Nonpharmacologic interventions are foundational to care and must be integrated early and maintained throughout the disease course rather than viewed as adjunctive. Although systemic corticosteroids remain the first-line standard, clearly defined timelines and objective criteria are essential to identify steroid-refractory or steroid-dependent disease and to prompt timely treatment escalation. The expanding availability of targeted agents—including BTK/ITK, JAK1/2, ROCK2, and CSF-1R inhibitors—now enables more precise therapeutic personalization based on organ involvement, immune phenotype, comorbidities, and prior treatment exposure. Ongoing, structured reassessment of disease activity, treatment intensity, and cumulative adverse event burden is critical to prevent both overtreatment and undertreatment, support durable remission, and ultimately move patients closer to immune tolerance.
References
- Flowers MED, Martin PJ. How we treat chronic graft-versus-host disease. Blood. 2015;125(4):606-615. https://doi.org/10.1182/blood-2014-08-551994
- Vadakkel G, Eng S, Proli A, Ponce DM. Updates in chronic graft-versus-host disease: novel treatments and best practices in the current era. Bone Marrow Transplant. 2024;59(10):1360-1368. https://doi.org/10.1038/s41409-024-02370-8
- Lee SJ, Zeiser R. FDA-approved therapies for chronic GVHD. Blood. 2025;145(8):795-800. https://doi.org/10.1182/blood.2024026633
- Ibrutinib (Imbruvica®) Prescribing information 2025. https://www.imbruvicahcp.com. Accessed 12/13/2025
- Pidala J, Kim J, Kalos D, et al. Ibrutinib for therapy of steroid-refractory chronic graft-versus-host disease: a multicenter real-world analysis. Blood Adv. 2025;9(5):1040-1048. https://doi.org/10.1182/bloodadvances.2024014374
- Cutler C, Lee SJ, Arai S, et al. Belumosudil for chronic graft-versus-host disease after 2 or more prior lines of therapy: the ROCKstar Study. Blood. 2021;138(22):2278-2289. https://doi.org/10.1182/blood.2021012021
- Lee SJ, Pavletic S, Blazar BR, et al. Belumosudil for chronic graft-versus-host disease: analysis of long-term results from the KD025-208 and ROCKstar studies. Transplant Cell Ther. 2025;31(7):434.e1-434.e10. https://doi.org/10.1016/j.jtct.2025.04.020
- Zeiser R, Polverelli N, Ram R, et al. Ruxolitinib for glucocorticoid-refractory chronic graft-versus-host disease. N Engl J Med. 2021;385(3):228-238. https://doi.org/10.1056/ NEJMoa2033122
- Mohty M, Socié G, Szer J, et al. Ruxolitinib versus best available therapy in patients with steroid-refractory acute graft-versus-host disease: final analysis from the randomized phase III REACH2 trial. J Clin Oncol. 2025;43(34):3639-3645. https://doi.org/10.1200/JCO-25-00809
- Wolff D, Cutler C, Lee SJ, et al. Axatilimab in recurrent or refractory chronic graft-versus-host disease. N Engl J Med. 2024;391(11):1002-1014. https://doi.org/10.1056/NEJMoa2401537